Unit 1: Structures, trends, chemical reactions, quantitative chemistry and analysis
Atomic structure
What are atoms made up of? How did scientists’ ideas about atoms change over time? What are protons, neutrons and electrons?

Bonding
What is the difference between ionic, covalent and metallic bonding? What are dot and cross diagrams? How are they interpreted?
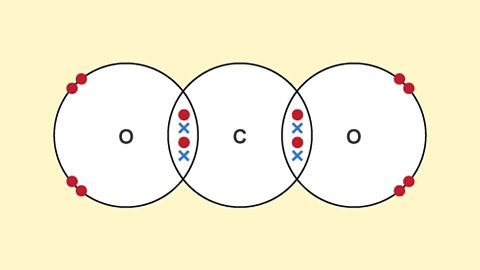
Structures
What are covalent and ionic compounds and structures? How do these two types of bonds lead to different physical properties in substances?

Nanoparticles
What are nanoparticles? What are they used for? What is their surface area to volume ratio?

Symbols, formulae and equations
What are symbols, formulae and equations? How do they help chemists explain chemical reactions in detail?

The periodic table
What is the history and structure of the periodic table? How do groups and periods work? How does reactivity change across the table?

Quantitative chemistry (1)
What are relative atomic masses? What are relative formula masses? How are these used in chemistry to carry out mole calculations?

Acids, bases and salts
How do indicators show whether a solution is acidic, alkaline or neutral? What makes acids and alkalis strong or weak? How do acids react with different substances?

Chemical analysis
How are chemicals analysed and separated using filtration; crystallisation; paper chromatography; simple and fractional distillation; and flame tests?

Solubility
What does solubility mean? How does temperature affect the solubility of solids and gases? What happens to a saturated solution when it is cooled?

Links
- External linkExternal link
- SubscriptionSubscription
- External linkExternal link
- External linkExternal link
- External linkExternal link
- SubscriptionSubscription
- External linkExternal link