Exam practice
GCSE Chemistry: CCEA past papers and mark schemes
Get ready for your chemistry exams with CCEA past papers and mark schemes, as well as top tips for your revision and exams in the subject.
GCSE Chemistry: exam-style quiz by topic
Try this quiz based on GCSE Chemistry past papers. Choose the topic you would like to revise and answer the questions.
GCSE Chemistry: exam-style questions
CCEA Foundation and higher GCSE interactive tests based on past papers to get you ready for your chemistry exams. Topics include the periodic table, equations and more.

GCSE Chemistry: quick-fire questions
Use our interactive quiz to understand how the CCEA foundation and higher chemistry GCSE exams work. Revise topics such as the periodic table and equations.

Quizzes
QUIZ: Acids, alkalis and salts (1)
This interactive quiz is for GCSE Chemistry (single science) students studying acids, alkalis and salts. Test your knowledge on neutralisation, reactions with acids and solutions.

QUIZ: Acids, alkalis and salts (2)
This interactive quiz is for GCSE Chemistry (single science) students studying acids, alkalis and salts. Test your knowledge of indicators, reactions and chemical equations.

Unit 1: Structures, trends, chemical reactions, quantitative chemistry and analysis
Atomic structure
What are atoms made up of? How did scientists’ ideas about atoms change over time? What are protons, neutrons and electrons?

Bonding
What is the difference between ionic, covalent and metallic bonding? What are dot and cross diagrams? How are they interpreted?
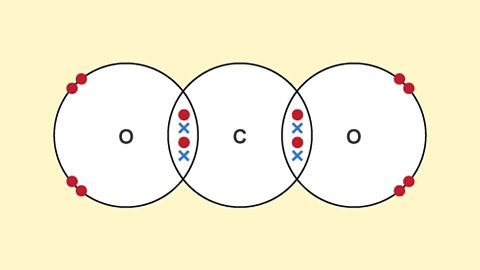
Structures
What are covalent and ionic compounds and structures? How do these two types of bonds lead to different physical properties in substances?

Nanoparticles
What are nanoparticles? What are they used for? What is their surface area to volume ratio?

Symbols, formulae and equations
What are symbols, formulae and equations? How do they help chemists explain chemical reactions in detail?

The periodic table
What is the history and structure of the periodic table? How do groups and periods work? How does reactivity change across the table?

Quantitative chemistry (1)
What are relative atomic masses? What are relative formula masses? How are these used in chemistry to carry out mole calculations?

Acids, bases and salts
How do indicators show whether a solution is acidic, alkaline or neutral? What makes acids and alkalis strong or weak? How do acids react with different substances?

Chemical analysis
How are chemicals analysed and separated using filtration; crystallisation; paper chromatography; simple and fractional distillation; and flame tests?

Solubility
What does solubility mean? How does temperature affect the solubility of solids and gases? What happens to a saturated solution when it is cooled?

Unit 2: Further chemical reactions, rates and equilibrium, calculations and organic chemistry
Metals and the reactivity series
What is the reactivity series? How does a more reactive metal displace a less reactive one from its compound? Why does reactivity decide how a metal can be extracted from its ore?

Redox, rusting and iron
Why does iron rust? How can rusting be prevented? What are redox reactions? How is iron extracted from its ore?

Rates of reaction
How do concentration, temperature, surface area and catalysts influence the rate of reaction? How is the rate of reaction be measured?

Equilibrium
How is dynamic equilibrium reached in a reversible reaction? What is Le Châtelier’s principle used to predict? What is the Haber process?

Organic chemistry
What are organic compounds? How are they grouped into homologous series like alkanes, alkenes, alcohols and carboxylic acids? How are they obtained from crude oil?

Quantitative chemistry (2)
How is concentration calculated from moles and volume? What is titration used for? What does Avogadro’s Law tell us about gas volumes? What is atom economy a measure of?

Electrochemistry
What occurs during electrolysis? How is an electrolyte split at the cathode and anode? How is aluminium extracted from its ore using electrolysis?

Energy changes in chemistry
What type of energy changes occur during chemical reactions? How are energy changes calculated? How does a reaction profile show these changes?
Gas chemistry
What type of gaseous elements and compounds are found in the Earth’s atmosphere? What are the key properties and uses of nitrogen, ammonia, hydrogen, oxygen and carbon dioxide?

Unit 3: Prescribed practicals
C1 - Determine the mass of water in hydrated crystals
Learn how to carry out this experiment where you heat hydrated iron(II) sulfate crystals, and record mass measurements, to calculate the mass of water in the supplied crystals.
C2 - Investigate the reactions of acids
Learn how to investigate the reactions of acids, including the temperature changes that occur.

C3 - Investigate the preparation of soluble salts
Learn how to make a soluble salt from a base and make a soluble salt from an alkali.

C4 - Identify the ions in an ionic compound
Learn how to identify the ions in an ionic compound using chemical tests.

C5 - Investigate the reactivity of metals
Learn how to determine the relative reactivity of metals through this experiment.

C6 - Investigate the rate of reaction
Learn about how to carry out an experiment to explore the rate of reaction.

C7 - Investigate the reactions of carboxylic acids
Learn how to carry out an experiment to investigate the reactions of carboxylic acids.

C8 - Titration
Determine the reacting volumes of solutions of acid and alkali by titration and determine the concentration of solutions of acid and alkali by titration.

C9 - Investigate the reaction of gases
Learn about how to investigate the preparation, properties, tests and reactions of the gases hydrogen, oxygen and carbon dioxide.

Practical skills
Practical skills
Plan, observe and evaluate experiments with a hypothesis, variables and conclusions, using scientific equipment like a measuring cylinder or a burette.

Links
- External linkExternal link
- SubscriptionSubscription
- External linkExternal link
- External linkExternal link
- External linkExternal link
- SubscriptionSubscription
- External linkExternal link