What are the key learning points about the structure of the nucleus?
What are the three particles inside the atomAll elements are made of atoms. An atom consists of a nucleus containing protons and neutrons, surrounded by electrons.?
The properties of protonSubatomic particle with a positive charge and a relative mass of 1. The relative charge of a proton is +1., electronA subatomic particle with relative mass of ¹⁄₁₈₄₀. The relative charge of an electron is -1. and neutronUncharged subatomic particle, with a relative mass of 1. The relative charge of a neutron is 0 (neutral)..
To understand atomic numberThe number of protons in the nucleus of an atom. and mass numberThe total number of protons and neutrons found in the nucleus of an atom..
What is an isotopeAtoms of an element with the same number of protons but different numbers of neutrons.?
What is the structure of the atom?
atomAll elements are made of atoms. An atom consists of a nucleus containing protons and neutrons, surrounded by electrons. are very small, they have a radius of around 0.1 nanometres
(1 × 10-10 metres).
The modern view of the atom is of a nucleus containing protonSubatomic particle with a positive charge and a relative mass of 1. The relative charge of a proton is +1. and neutronUncharged subatomic particle, with a relative mass of 1. The relative charge of a neutron is 0 (neutral). with electronA subatomic particle with relative mass of ¹⁄₁₈₄₀. The relative charge of an electron is -1. orbiting around the nucleus in shellAn energy level around the nucleus where electrons can be found orbiting..
The nucleusThe central part of an atom. It contains protons and neutrons, and has most of the mass of the atom. The plural of nucleus is nuclei. is tiny compared to the overall size of the atom, (about 10000 times smaller).
The table below summarises the important information about each particle.
| Particle | Relative mass | Relative charge | Location |
|---|---|---|---|
| Proton | 1 | +1 | In the nucleus |
| Neutron | 1 | 0 (neutral) | In the nucleus |
| Electron | \(\frac{1}{1840}\) | -1 | Orbiting the nucleus in shells |
Key facts
Protons and neutrons are the heaviest particles in an atom and as a result they make up most of its mass.
The mass of electrons is very small and often not considered to be significant.
An atom is always electrically neutral (i.e. has no overall charge) as the number of positive protons equals the number of negative electrons.
If the atom becomes ionisationProcess by which electrons can be added or removed from an atom to create an ion. however, the number of electrons will change.
An ion is an atom that has lost or gained one or more electron.
Mass number A and atomic number Z
The number of protons is called the atomic number Z.
The number of protons is what defines the element, i.e. an atom with six protons in its nucleus will always be carbon, and uranium will always have 92 protons.
The total number of particles in the nucleus, (i.e. the sum of all the protons and neutrons), is called the mass number A.
What makes up the structure of the nucleus?
Mass number and atomic number are two important pieces of information about a nucleus.
A nucleus can be represented using the symbol notation:
\(_{Z}^{A}\textrm{X}\)
Where:
- A is the mass number (the sum of the protons and neutrons)
- Z is the atomic number (the number of protons)
- X is the chemical symbol of the element
Note: the number of neutrons in the nucleus can be calculated by subtracting Z from A.
For example, chlorine (Cl) can be shown as:
What are isotopes?
Cl is the chemical symbol for chlorine.
A chlorine nucleus will contain:
- Number of protons Z = 17
- Number of protons + neutrons A = 35
- Number of neutrons N = 35 - 17 = 18
The chlorine atom will also have 17 electrons, as an atom is neutral.
Remember, there are no electrons in the nucleus.
The atomic number of an atom determines which element it is.
An element with 17 protons will always be chlorine.
However the mass number of the element can vary, which means that it can have different numbers of neutrons.
Although chlorine always has 17 protons, two different atoms of chlorine can have different numbers of neutrons.
The different types of chlorine are called isotopes of chlorine.
Isotopes are different atoms of the same element. They have the same number of protons but different numbers of neutrons.
This means the isotopes will have the same atomic number but a different mass number.
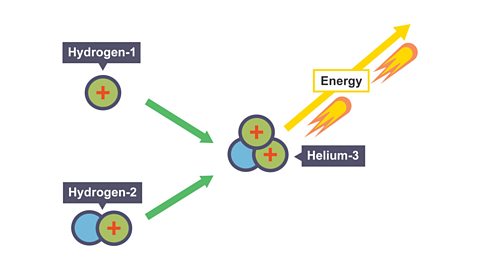
There are three isotopes of hydrogen: hydrogen, deuterium (hydrogen-2) and tritium (hydrogen-3):
Carbon has three isotopes: \(_{6}^{12}\textrm{C}\), \(_{6}^{13}\textrm{C}\) and \(_{6}^{14}\textrm{C}\).
They all contain six protons, but six, seven and eight neutrons respectively.
\(_{7}^{14}\textrm{N}\) and \(_{6}^{14}\textrm{C}\) are not isotopes as they are different elements (they have a different number of protons in the nucleus).
They have the same mass number, A, but different atomic number, Z, and different chemical symbol - they are not the same element.
If the number of protons changes, then it is a different element.
Question
How many protons does a \(_{6}^{14}\textrm{C}\) atom contain?
Answer
The atomic number is 6 so \(_{6}^{14}\textrm{C}\) contains six protons.
Question
How many neutrons are in the nucleus of a \(_{6}^{14}\textrm{C}\) atom?
Answer
Number of neutrons = mass number - atomic number = 14 - 6 = 8 neutrons.
Question
How many electrons are in the nucleus of a \(_{6}^{14}\textrm{C}\) atom?
Answer
There are no electrons in the nucleus.
Question
Write down the number of particles in the following nuclei:
\(_{30}^{64}\textrm{Zn}\)
\(_{92}^{238}\textrm{U}\)
\(_{1}^{3}\textrm{H}\)
Answer
\(_{30}^{64}\textrm{Zn}\)
- Number of protons Z = 30
- Number of protons + neutrons A = 64
- Number of neutrons N = (64 – 30) = 34
- Since this is a nucleus there are no electrons
There are 30 protons and 34 neutrons.
\(_{92}^{238}\textrm{U}\)
- Number of protons Z = 92
- Number of protons + neutrons A = 238
- Number of neutrons N = 238 - 92 = 146
- Since this is a nucleus there are no electrons
There are 92 protons and 146 neutrons.
\(_{1}^{3}\textrm{H}\)
- Number of protons Z = 1
- Number of protons + neutrons A = 3
- Number of neutrons N = 3 - 1 = 2
- Since this is a nucleus there are no electrons
There is 1 proton and 2 neutrons.
Question
A helium nucleus 4He has 2 neutrons. How many protons are there?
Answer
The number of protons + neutrons A = 4.
If the number of neutrons N is 2 then there must be 4 - 2 = 2 protons.
There are 2 protons in the helium nucleus.
What is ionisation?
atomAll elements are made of atoms. An atom consists of a nucleus containing protons and neutrons, surrounded by electrons. are electrically neutral.
This means that overall they are not positive or negative.
They have the same number of positive protons in the nucleus as they have negative electrons orbiting in the energy levels around the nucleusThe central part of an atom. It contains protons and neutrons, and has most of the mass of the atom. The plural of nucleus is nuclei..
When an atom loses or gains an electron, it forms a charged particle called an ionElectrically charged particle, formed when an atom gains or loses electrons.:
If the atom loses one or more electrons, it will have more protons than electrons and so it becomes a positively-charged ion.
If the atom gains one or more electrons, it will have more electrons than protons and so it becomes a negatively-charged ion.
The atom will not normally gain or lose protons because they are held very tightly inside the nucleus. Electrons are much more loosely held.
Key fact
Ionisation is the addition or removal of an electron to create an ion.
A helium atom has two electrons orbiting the nucleus.
The atom is neutral as it has two positive protons and two negative electrons.
A helium atom that has lost or gained an electron is a helium ion.
The ion has two positive protons but one electron so it is a positive ion:
The ion has two positive protons but three electrons so it is a negative ion:
Question
Describe in detail the present model of the atom known as the Rutherford-Bohr model.
Name the particles that make up the atom, their mass and their location.
Question
| Relative charge | Relative mass | Location | |
|---|---|---|---|
| Proton | +1 | 1 | Inside the nucleus |
| Neutron | 0 | 1 | Inside the nucleus |
| Electron | -1 | \(\frac{\text{1}}{\text{1840}}\) | Orbiting the nucleus |
Question
The element of potassium has the symbol K.
The nucleus of an atom of potassium contains 19 protons and 20 neutrons.
Using this information complete the symbol below:
\(_{?}^{?}\textrm{K}\)
There are 19 protons and so atomic number Z = 19.
There are 19 protons and 20 neutrons and so the mass number A = 19 + 20 = 39.
\(_{19}^{39}\textrm{K}\)
Question
The element sodium has the symbol \(_{11}^{23}\textrm{Na}\).
How many protons, neutrons and electrons can be found in the nucleus of a sodium atom?
Answer
The atomic number Z = 11 and so there are 11 protons.
The mass number A = 23.
The number of neutrons N = A - Z = 23 - 11 = 12 neutrons.
There are no electrons in the nucleus of a sodium atom.
There are 11 protons, 12 neutrons and 0 electrons inside a sodium nucleus.
Test your knowledge
More on Unit 1: Atomic and nuclear physics
Find out more by working through a topic
- count3 of 6

- count4 of 6
- count5 of 6
- count6 of 6